

Extending expert Oncology Clinical Trial Services in the USA, India, and Europe, covering all oncology clinical trial phases with regulatory-compliant clinical trial design, well-defined clinical trial endpoints, compliant protocol development, regulatory support, and patient-focused solutions.
An oncology clinical trial is a structured research study designed to evaluate new treatments, drugs, combinations, or approaches for the prevention, diagnosis, or treatment of cancer. These studies follow a predefined protocol and progress through different oncology clinical trial phases to assess safety, dosage, efficacy, and side effects, with clearly defined clinical trial endpoints such as tumor response, survival rates, and quality of life to measure outcomes under strict regulatory and ethical guidelines.
Building on these scientific and regulatory principles, ProRelix Research, a leading CRO, offers oncology clinical trial services in the USA, India, and Europe. With expertise in protocol development, global regulatory compliance, and patient-focused trial execution, ProRelix Research supports high-quality oncology studies across all trial phases.
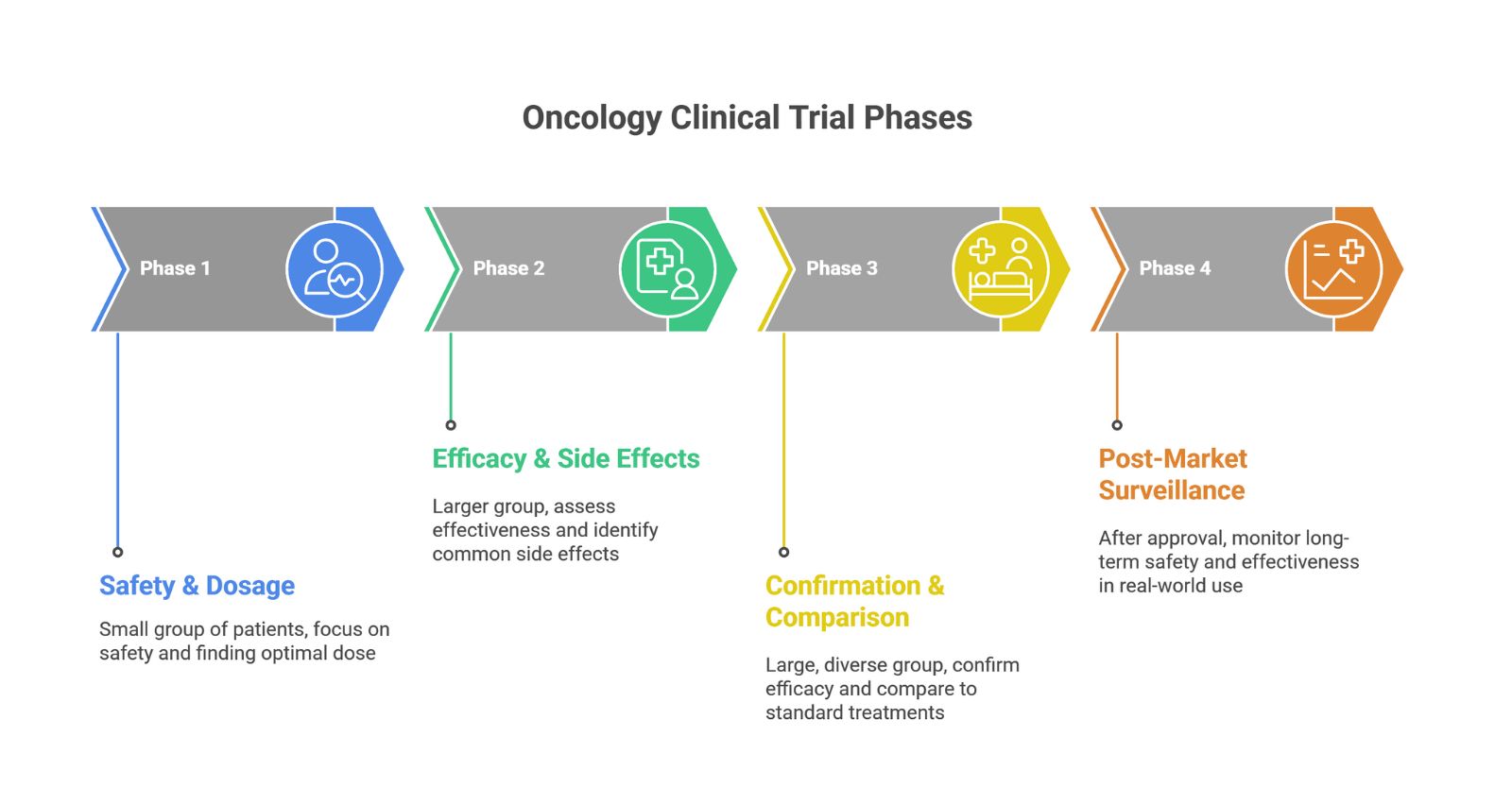
Oncology clinical trials are conducted in sequential phases to ensure the safety, effectiveness, and reliability of cancer treatments, ProRelix Research provides comprehensive Oncology Clinical Trials Phases support, helping sponsors successfully plan, manage, and execute cancer studies from early development to post-marketing under global regulatory standards.
In Phase 0 clinical trials, very small doses of an investigational drug are used to study its biological activity, target interaction, and pharmacokinetics before advancing to larger studies. These early findings help determine whether the therapy should move forward into formal clinical development.
Phase 1 clinical trials focus on evaluating safety, determining dosage ranges, and identifying side effects in a small group of patients with cancer. This phase establishes the maximum tolerated dose and appropriate administration schedule.
During Phase 2 clinical trials, researchers assess preliminary treatment effectiveness, further evaluate safety, and refine dosing in a larger patient population. Results from this phase help confirm whether the therapy shows sufficient clinical benefit to proceed.
Phase 3 clinical trials compare the new oncology treatment with the current standard of care to confirm efficacy, monitor adverse reactions, and support regulatory approval. These trials provide the pivotal data required for marketing authorization submissions.
In Phase 4 clinical trials, ongoing studies are conducted after approval to monitor long-term safety, effectiveness, and real-world clinical outcomes. This phase helps identify rare side effects and supports continuous improvement of cancer therapies.

Well-planned oncology trial design and clear protocols ensure meaningful endpoints, reliable data, and faster decision-making.
Strong regulatory alignment keeps oncology trials compliant, audit-ready, and on track for global approvals.
Focused patient recruitment, engagement, and safety monitoring drive smoother trials and better outcomes.
Cancer clinical trials are classified based on their purpose and the stage of research. The main types include:
Test new cancer treatments such as drugs, immunotherapy, targeted therapy, radiation, surgery, or combination therapies.
Study ways to reduce cancer risk using medicines, vaccines, or lifestyle interventions.
Examine genetic markers to predict cancer risk, treatment response, or disease progression.
Evaluate new tests or procedures to detect cancer earlier or more accurately.
Assess methods to identify cancer before symptoms appear.
Focus on reducing side effects, managing symptoms, and improving patient comfort during or after cancer treatment.
Oncology clinical trial design is the structured planning process used to evaluate the safety, efficacy, and therapeutic value of cancer treatments. It defines the study objectives, patient eligibility criteria, treatment arms, dosing strategy, and statistical methodology. Effective oncology clinical trial design incorporates clearly defined clinical trial endpoints such as tumor response, progression-free survival, and overall survival. It also accounts for biomarker strategies, adaptive designs, and ethical considerations to ensure patient safety. A well-designed oncology trial improves data quality, accelerates decision-making, and supports successful regulatory submissions.
Building on these principles, ProRelix Research provides Oncology Clinical Trial Design, offering end-to-end support from protocol development to regulatory alignment to ensure high-quality, patient-centric oncology studies.

Oncology clinical trial endpoints are specific measurable outcomes used to evaluate the effectiveness and safety of cancer treatments. They provide clear criteria for assessing how well a therapy works and guide decisions throughout the trial. Common endpoints include overall survival (OS), progression-free survival (PFS), tumor response rates, disease-free survival, and quality of life measures. Selecting the right endpoints is crucial for trial design, regulatory approval, and demonstrating clinical benefit. Well-defined endpoints ensure meaningful, reliable data while keeping patient safety and treatment goals at the forefront.
ProRelix Research provides Oncology Clinical Trial Endpoints support, helping sponsors define, implement, and monitor precise endpoints to achieve robust, regulatory-compliant oncology study outcomes.
Overall survival measures the time from the start of treatment until death from any cause. It is considered the gold standard endpoint in oncology trials, reflecting the ultimate clinical benefit of a therapy.
Progression-free survival tracks the length of time during and after treatment that a patient lives without cancer worsening. It helps evaluate the effectiveness of a therapy in controlling disease.
Disease-free survival measures the period after treatment during which a patient shows no signs of cancer recurrence. It is commonly used in adjuvant therapy trials to assess long-term treatment success.
This endpoint quantifies the proportion of patients whose tumors shrink or disappear after treatment. It provides an early measure of a therapy’s anti-cancer activity.
Time to progression measures how long it takes for a patient’s cancer to worsen after starting treatment. Unlike PFS, it does not consider death as an event unless related to cancer progression.
Duration of response tracks how long a patient maintains a positive tumor response before disease progression. It helps determine the lasting impact of a therapy.
Quality of life assesses the patient’s physical, emotional, and social well-being during and after treatment. It is a critical endpoint to evaluate tolerability and patient-centered benefits.
Minimal residual disease measures the small number of cancer cells that remain after treatment, often used in hematologic malignancies to predict relapse risk.
Event-free survival measures the time from treatment start until a defined event occurs, such as disease progression, recurrence, or death. It helps assess overall treatment effectiveness.
Monitoring safety and adverse events evaluates the type, frequency, and severity of side effects from a treatment. This ensures patient safety and informs risk-benefit decisions.
An oncology clinical trial protocol is a detailed plan that outlines the objectives, design, methodology, and operational procedures of a cancer study. It serves as a roadmap for conducting the trial, ensuring consistency, scientific rigor, and patient safety. A well-prepared protocol defines the study population, treatment regimens, dosing schedules, inclusion and exclusion criteria, clinical trial endpoints, and statistical analysis methods. It also specifies safety monitoring, reporting requirements, and ethical considerations to comply with regulatory standards. A strong protocol is critical for trial success, helping investigators generate reliable, high-quality data and supporting regulatory submissions.
ProRelix Research provides Oncology Clinical Trial Protocol development services, offering end-to-end support to design scientifically robust, regulatory-compliant, and patient-focused oncology studies.
Overall survival measures the time from the start of treatment until death from any cause. It is considered the gold standard endpoint in oncology trials, reflecting the ultimate clinical benefit of a therapy.
Progression-free survival tracks the length of time during and after treatment that a patient lives without cancer worsening. It helps evaluate the effectiveness of a therapy in controlling disease.
Disease-free survival measures the period after treatment during which a patient shows no signs of cancer recurrence. It is commonly used in adjuvant therapy trials to assess long-term treatment success.
This endpoint quantifies the proportion of patients whose tumors shrink or disappear after treatment. It provides an early measure of a therapy’s anti-cancer activity.
Time to progression measures how long it takes for a patient’s cancer to worsen after starting treatment. Unlike PFS, it does not consider death as an event unless related to cancer progression.
Duration of response tracks how long a patient maintains a positive tumor response before disease progression. It helps determine the lasting impact of a therapy.
Quality of life assesses the patient’s physical, emotional, and social well-being during and after treatment. It is a critical endpoint to evaluate tolerability and patient-centered benefits.
Minimal residual disease measures the small number of cancer cells that remain after treatment, often used in hematologic malignancies to predict relapse risk.
Event-free survival measures the time from treatment start until a defined event occurs, such as disease progression, recurrence, or death. It helps assess overall treatment effectiveness.
Monitoring safety and adverse events evaluates the type, frequency, and severity of side effects from a treatment. This ensures patient safety and informs risk-benefit decisions.
Oncology Clinical Trials and Therapeutics focus on advancing cancer treatment through scientifically designed research studies that evaluate new drugs, biologics, immunotherapies, targeted therapies, and combination approaches. These trials follow structured oncology clinical trial phases, supported by clear protocols, strategic clinical trial design, and measurable clinical trial endpoints such as tumor response, progression-free survival, and overall survival. By integrating innovative therapeutic strategies with regulatory and ethical standards, oncology clinical trials help determine safety, dosage, efficacy, and long-term outcomes. Patient-focused methodologies and continuous safety monitoring ensure trial integrity and reliable results. ProRelix Research specializes in Oncology Clinical Trials and Therapeutics, offering end-to-end clinical research support aligned with global regulatory standards and patient-centric execution worldwide for oncology innovation.

We offer comprehensive support across all oncology clinical trials phases, from Phase 1 to Phase 4, covering drug, biologic, and therapeutic development.
Key factors in oncology clinical trial design include defining treatment arms, patient population, sample size, randomization methods, clinical trial endpoints, and statistical analysis plans. Other considerations include adaptive design options, biomarker strategies, ethical compliance, and patient safety monitoring. Proper trial design ensures reliable, actionable results while minimizing risks and maximizing efficiency for sponsors and patients.
The most common oncology clinical trial endpoints are measurable outcomes used to evaluate the effectiveness and safety of cancer therapies. These include overall survival (OS), progression-free survival (PFS), tumor response rate (TRR), disease-free survival (DFS), and quality of life (QoL). Selecting the right endpoints ensures the trial generates meaningful, regulatory-compliant data and provides actionable insights for treatment decisions.
An oncology clinical trial protocol is a detailed blueprint that guides every aspect of a cancer study. It defines the study objectives, patient eligibility, treatment regimens, endpoints, safety monitoring, and statistical methods. A well-developed protocol ensures regulatory compliance, patient safety, and high-quality data collection, enabling smooth trial execution and reliable results for regulatory submissions.
Eligibility for cancer clinical trials depends on factors such as cancer type, stage, prior treatments, overall health, and specific inclusion and exclusion criteria defined in the oncology clinical trial protocol.
Current clinical trials for cancer patients include studies on immunotherapy, targeted therapy, precision medicine, combination treatments, and advanced biologics. These trials are conducted worldwide across multiple cancer types and stages.
The success of cancer clinical trials depends on the trial phase, cancer type, and treatment approach. Early-phase trials focus on safety, while later phases evaluate effectiveness. Many approved cancer therapies today are the result of successful oncology clinical trials.
ProRelix Research provides comprehensive Oncology Clinical Trial Services in the USA, India, and Europe. With expertise in clinical trial design, protocol development, regulatory compliance, patient-centric execution, and global trial management, ProRelix Research helps sponsors conduct high-quality, efficient, and compliant oncology studies across all phases.
If you are planning to develop novel treatments in any therapeutic area, it’s time to consult a CRO that delivers rigor, efficiency, and regulatory clarity at every stage. Let’s position your therapy for global markets.

Be the first to know the latest trends in clinical research, real-world case studies, and industry secrets.

ProRelix Research is the rapidly growing Contract/ Clinical Research Organization (CRO) with multi-country service capability supporting phase 1, 2, 3, & 4 clinical trials of Pharma, Biotech, Biopharma, Medical Device, Nutraceutical & Herbal companies to conduct in the USA, India, Europe & South East Asia.