

ProRelix Research is a premier preclinical Contract Research Organization (CRO) in the USA, India, and Europe, offering comprehensive CRO solutions that include study design, protocol development, data management, analysis, and regulatory support.
Sponsors today encounter growing complexities throughout clinical development as they navigate slow patient enrollment, protocol deviations, inconsistent site performance, and high screen-failure variations. Many studies struggle with delayed site activations, substantial data query backlogs, and challenges related to inspection readiness. Global projects further intensify these issues with country-specific regulatory expectations, logistical constraints, cost escalation, and the ongoing need to maintain uniform data quality across diverse geographies. To overcome these challenges, sponsors require scientifically driven and operationally experienced partners capable of anticipating risks early and guiding programs with strategic precision.
ProRelix Research is a High-Performance CRO providing complete Contract Research Organization services, committed to advancing clinical development through scientifically robust, compliant, and efficiently executed solutions. We collaborate with global sponsors to design strategic study frameworks, oversee end-to-end clinical trial operations, and ensure excellence across regulatory affairs, pharmacovigilance, data management, and site management activities. With deep industry expertise and a strong quality-driven mindset, ProRelix Research empowers sponsors to accelerate innovation, optimize trial integrity, and achieve successful outcomes in an increasingly complex clinical research landscape.
A Contract Research Organization (CRO) provides end-to-end support to pharmaceutical, biotechnology, and medical device companies by managing the entire spectrum of clinical development. This includes designing study protocols, overseeing clinical trial operations, managing data collection and analysis, ensuring regulatory compliance, conducting site management, and maintaining pharmacovigilance. CROs act as strategic partners, enabling sponsors to accelerate timelines, optimize resources, maintain high-quality standards, and navigate the complexities of global clinical research with scientific precision and operational excellence.
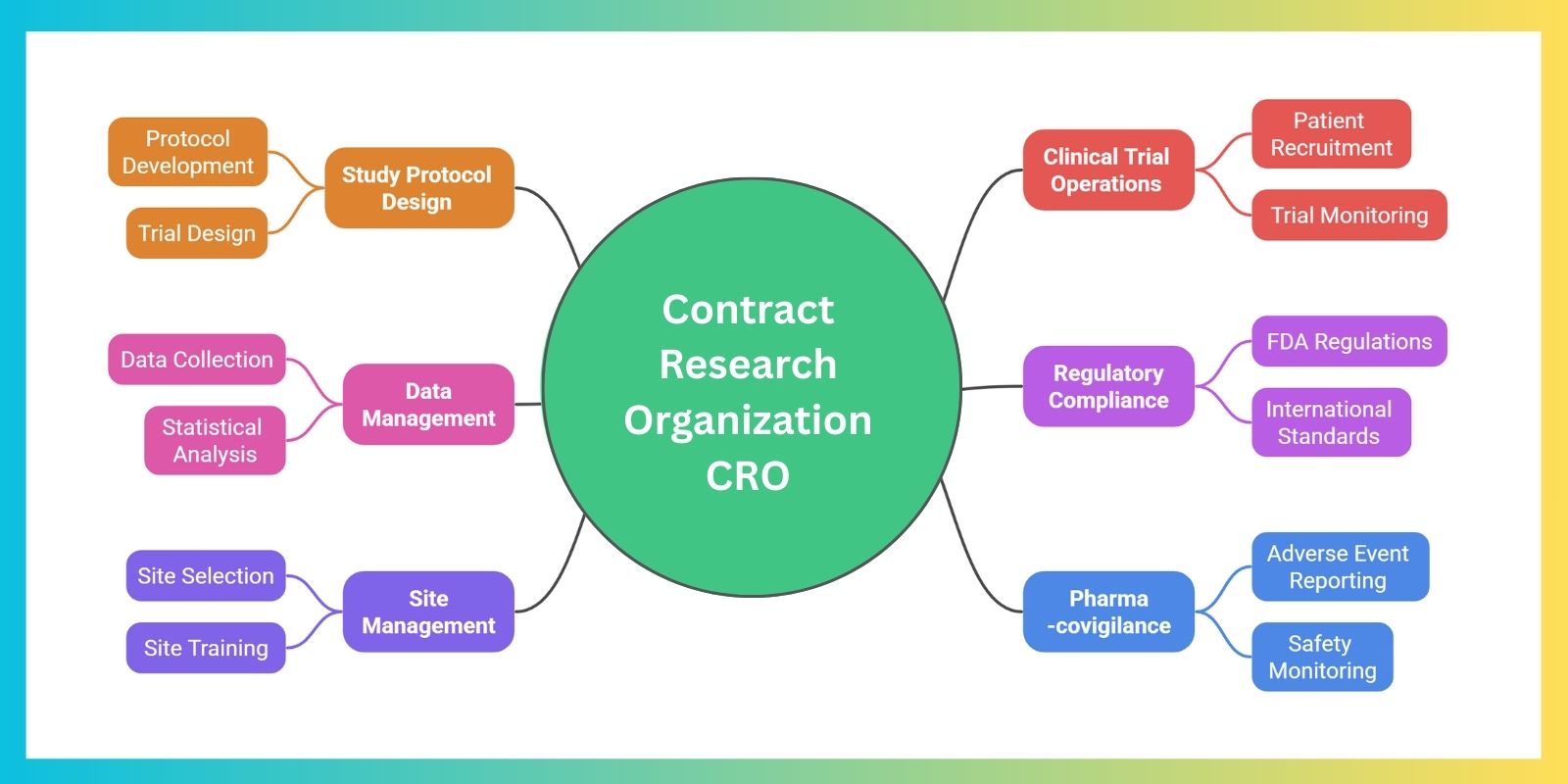
As a leading clinical research organization, ProRelix Research provides end-to-end contract research organization services that help sponsors overcome operational and regulatory challenges. In a rapidly expanding contract research organization market size, our expertise ensures efficient trial execution, high-quality data, and reliable study outcomes.
By combining scientific expertise with innovative trial management approaches, ProRelix Research reduces operational risks, enhances data quality, and accelerates decision-making. Our collaborative model empowers sponsors to make informed choices, achieve faster milestones, and maintain a competitive edge in an evolving clinical development environment.
Beyond the clinical clouds, far from the static pages, lies our portfolio, where each project is a spark of progress and possibility.
Beyond the data valleys, far from the protocols of Compliance and Auditland, our experts conduct Phase 1 to 4 trials with precision and care. Trusted processes thrive in Clinical Haven.
Across the operational plains, far from the delays of logistics and hurdles, our teams manage sites with seamless coordination. Consistency lives in TrialStream Valley.

Among the language peaks, beyond the lands of confusion and drafts, our writers craft clarity from complex data. Structured words dwell in Reportsville Fields.
Within the digital forests, away from the noise of raw entries and missing fields, our team ensures clean, validated data. Accuracy blooms in Dataset Grove.
The contract research organization market is growing rapidly as pharmaceutical, biotechnology, and medical device companies increasingly outsource clinical trial operations to improve efficiency, reduce costs, and access global expertise. The overall contract research organization market size continues to expand due to rising R and D investments, a surge in complex clinical studies, and strong demand for specialized services. Within Europe, the France contract research organization market and the Germany contract research organization market stand out as key contributors, supported by mature healthcare infrastructures, strong regulatory systems, and high clinical trial activity.
As the market expands, ProRelix Research is strategically leveraging this growth by strengthening its global delivery capabilities and offering end to end clinical trial support tailored to sponsor needs. With the increasing market size, ProRelix Research is able to collaborate with more international clients, expand its therapeutic expertise, and deliver faster, more cost effective study solutions. This growing demand allows the organization to scale operations, adopt advanced technologies, and position itself as a trusted partner for companies seeking reliable and efficient CRO services.
Projects Completed
Site Network
Regulatory Authorities' Experience
Years in Business

A contract research organization (CRO) is a service provider that supports pharmaceutical, biotechnology, and medical device companies in managing clinical trials, regulatory compliance, and research activities. CROs help sponsors accelerate development while maintaining data quality and operational efficiency.
The best CRO depends on the study type, therapeutic area, and sponsor requirements. Leading organizations are recognized for scientific expertise, regulatory knowledge, and proven track records in successful clinical trial management.
Selecting the right CRO involves evaluating experience in the relevant therapeutic area, regulatory compliance record, operational efficiency, and global capabilities. ProRelix Research is an example of a partner that combines these strengths with a collaborative approach to deliver tailored solutions.
Leading multinational CROs like IQVIA, ICON, Covance, and Parexel manage extensive global trial portfolios across multiple therapeutic areas.
CROs design, manage, and monitor clinical trials while providing regulatory, data management, and pharmacovigilance support. They ensure trials are executed efficiently, safely, and in compliance with global standards.
A CRO FDA refers to a CRO operating under FDA regulations, ensuring compliance with Good Clinical Practice and other federal requirements. These CROs support sponsors in meeting U.S. regulatory standards for drug and device approvals.
CROs provide end-to-end clinical development services including study planning, site management, data analysis, and regulatory submissions. ProRelix Research delivers these services with scientific rigor and operational excellence, helping sponsors achieve timely and reliable trial outcomes.
A global CRO manages international clinical trials while navigating diverse regulatory and operational environments. These organizations provide sponsors with scalable expertise, local knowledge, and efficient cross-border trial execution.
Some of the top global Contract Research Organizations include IQVIA, ICON plc, Parexel, ProRelix Research, PPD (Thermo Fisher Scientific), Labcorp Drug Development (Covance), Syneos Health, Medpace, Charles River Laboratories, WuXi AppTec, and Premier Research.
Be the first to know the latest trends in clinical research, real-world case studies, and industry secrets.

ProRelix Research is the rapidly growing Contract/ Clinical Research Organization (CRO) with multi-country service capability supporting phase 1, 2, 3, & 4 clinical trials of Pharma, Biotech, Biopharma, Medical Device, Nutraceutical & Herbal companies to conduct in the USA, India, Europe & South East Asia.