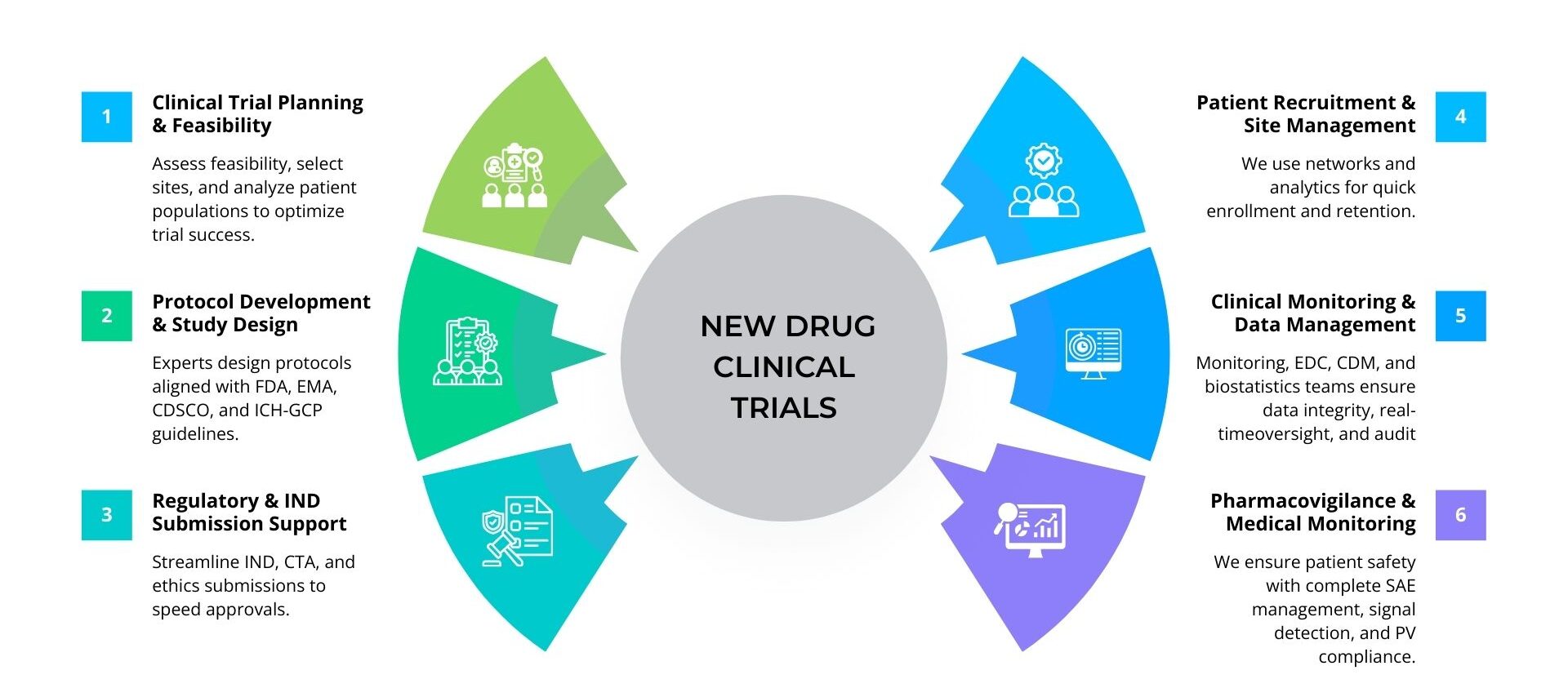
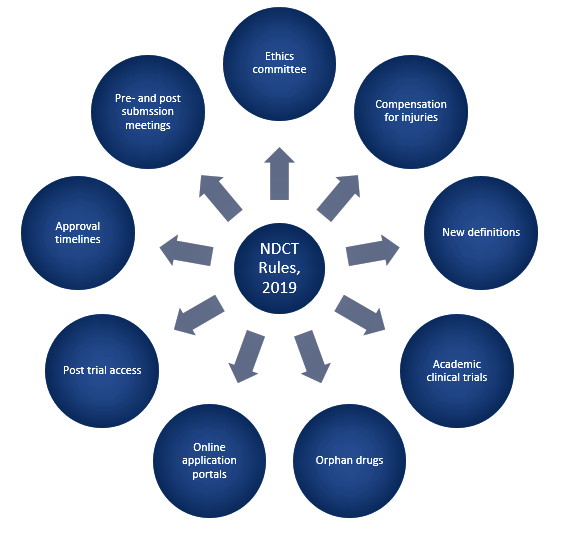
Our team of experts supports sponsors across every stage of new drug development by designing robust clinical trials, preparing accurate regulatory submissions, and ensuring continuous compliance with evolving global guidelines, making ProRelix Research a trusted CRO for bringing new drugs to market efficiently and responsibly. We proactively monitor regulatory updates from authorities such as CDSCO, US FDA, EMA, and ICH to keep clinical trial processes fully aligned with the latest requirements, covering everything from Investigational New Drug (IND) applications and protocol amendments to safety reporting, audits, and inspections. This compliance-driven approach minimizes regulatory risks, prevents delays, ensures subject protection and transparency, and enables smoother approvals throughout the entire new drug clinical trial lifecycle.