

Advancing clinical research for metabolic and hormonal disorders through scientifically robust, patient-focused studies. ProRelix Research provides end-to-end endocrinology & diabetology clinical trials support aligned with global regulatory standards.
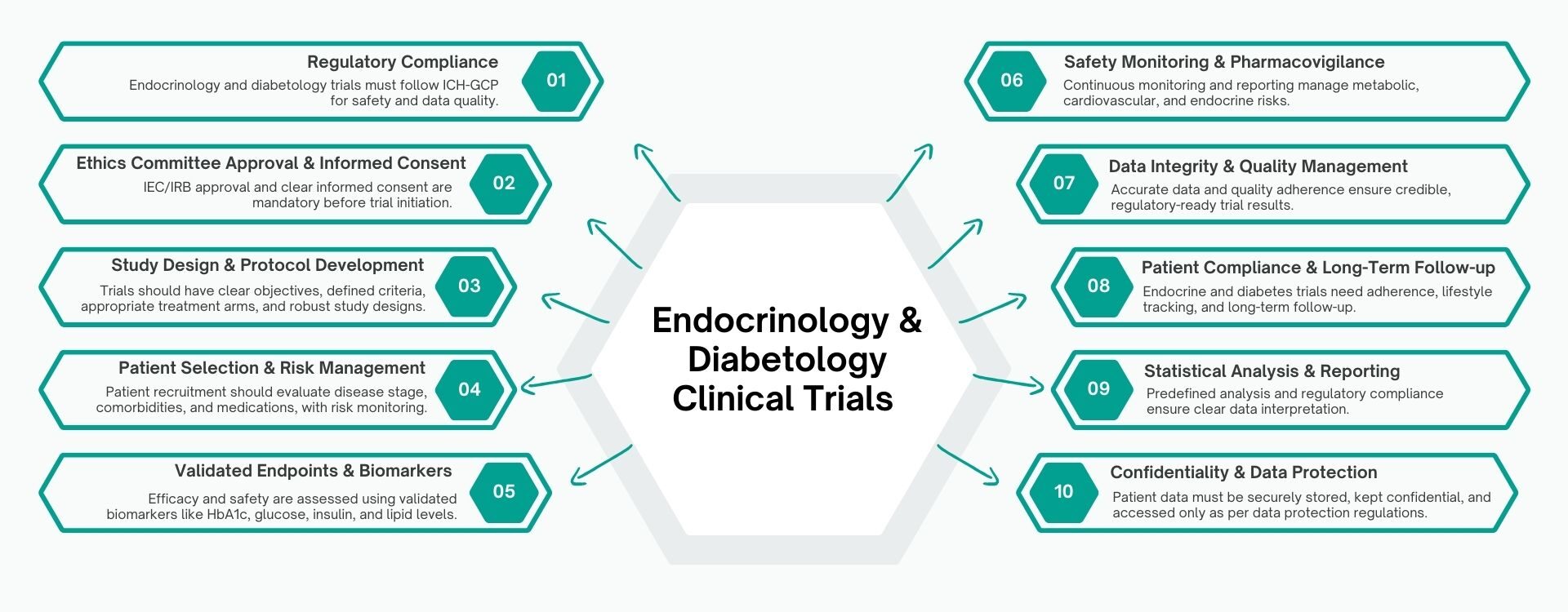
Endocrinology & Diabetology clinical trials advance therapies for metabolic and hormonal disorders through studies evaluating drugs, biologics, insulin delivery systems, and combination treatments. Following structured trial phases with robust protocols and endpoints such as glycemic control, hormone balance, and metabolic improvement, these trials integrate innovative therapies with strict regulatory and ethical standards to ensure safety, efficacy, and long-term patient outcomes. ProRelix Research provides global endocrinology & diabetes clinical trial support, delivering end-to-end, regulatory-compliant, and patient-focused research solutions to drive innovation in metabolic health worldwide.
Clinical Endocrinology is a medical specialty focused on the diagnosis, treatment, and management of disorders of the endocrine system, which regulates hormones responsible for metabolism, growth, reproduction, and overall body function. It addresses conditions such as diabetes, thyroid disorders, obesity, osteoporosis, adrenal and pituitary diseases, and other hormonal imbalances through hormone testing, imaging, and metabolic assessments. Clinical endocrinology applies evidence-based therapies, including medications, hormone replacement, and long-term disease management strategies, to support effective control of endocrine disorders and improve patient outcomes
Endocrinology Clinical Practice guidelines provide standardized approaches that guide clinical research. Endocrinology & Diabetology clinical trials follow strict scientific, ethical, and regulatory standards to ensure patient safety, data integrity, and reliable outcomes, focusing on disorders like diabetes, thyroid diseases, obesity, adrenal disorders, and hormonal imbalances.
Diabetes clinical trials are designed to evaluate new treatments, technologies, and care approaches for different forms of diabetes. These trials help improve disease management, reduce complications, and enhance patient quality of life.
These trials focus on autoimmune-related diabetes, where the body’s immune system attacks insulin-producing beta cells. Studies may evaluate immune-modulating therapies, beta-cell preservation strategies, insulin alternatives, and artificial pancreas systems to improve glucose control and delay disease progression.
Type 2 diabetes trials assess new oral medications, injectable therapies, lifestyle interventions, and combination treatments. These studies aim to improve insulin sensitivity, enhance insulin secretion, support weight management, and reduce cardiovascular and metabolic risks associated with the condition.
These trials involve diabetes that develops during pregnancy. Research focuses on safe and effective glucose-lowering therapies, dietary and lifestyle interventions, and maternal-fetal outcome improvements to reduce risks for both mother and baby.
Prediabetes studies aim to prevent or delay the progression to Type 2 diabetes. Trials often evaluate lifestyle modification programs, preventive medications, digital health tools, and early intervention strategies to improve insulin resistance and glucose tolerance.
These trials assess insulin pumps, continuous glucose monitoring (CGM) systems, smart insulin pens, and digital health platforms. The goal is to improve real-time glucose tracking, treatment adherence, and overall diabetes management through innovative technologies.
These studies focus on preventing or treating diabetes-related complications such as neuropathy, nephropathy, retinopathy, and cardiovascular disease. Trials evaluate therapies that protect organs, improve long-term outcomes, and reduce diabetes-related morbidity

Endocrine & diabetes clinical trials advance the understanding and treatment of metabolic and hormonal disorders. They follow structured protocols ensuring patient safety, data integrity, and regulatory compliance, addressing conditions like diabetes, thyroid, obesity, adrenal, and pituitary disorders.
Clearly defined primary and secondary objectives, including glycemic control, hormone regulation, weight management, or biomarker changes.
Randomized, controlled, observational, or interventional study frameworks tailored to disease type and therapy.
Inclusion and exclusion criteria that define the target population, considering age, disease stage, comorbidities, and prior therapies.
Detailed description of interventions, dosage schedules, administration routes, and duration of therapy.
Regular clinical evaluations, laboratory testing, adverse event monitoring, and compliance with endocrinology clinical practice guidelines.
Standardized procedures for capturing, storing, and analyzing patient data to maintain accuracy and confidentiality.
Informed consent, patient privacy, and adherence to regulatory requirements such as ICH-GCP guidelines.
Predefined methods to evaluate efficacy and safety outcomes, including handling of missing data and interim analyses.
Clinical research in diabetes & endocrinology plays a important role in understanding metabolic disorders, hormonal imbalances, and related chronic conditions. High-quality studies in this field not only advance clinical knowledge but also guide evidence-based practice and policy-making.
The impact factor of diabetes & endocrinology research studies reflects the significance and influence of these publications in the scientific community. Journals with higher impact factors are recognized for publishing groundbreaking findings, robust clinical trials, and innovative therapies that shape patient care standards. Many of these studies are conducted through established Diabetes Clinical Trial networks, ensuring standardized protocols, patient safety, and high-quality data collection.
Tracking the impact factor of relevant journals helps researchers, clinicians, and institutions identify influential studies, prioritize high-quality evidence, and align ongoing research with global scientific standards. It also ensures that clinical guidelines and therapeutic decisions are informed by the most credible and widely cited research in the field of endocrinology & metabolic health.
Clinical research in Endocrinology & Diabetes & Metabolism focuses on advancing the understanding, prevention, and management of complex hormonal and metabolic disorders, including diabetes mellitus, obesity, thyroid and adrenal disorders, lipid abnormalities, osteoporosis, and rare metabolic conditions. ProRelix Research brings expertise in scientifically robust study design, clearly defined metabolic and glycemic endpoints, and protocols tailored to chronic and progressive diseases, ensuring patient safety, regulatory compliance, and clinically meaningful outcomes. Integration of biomarkers, digital health tools, and long-term assessments supports high-quality, real-world evidence across all phases of clinical trials in Endocrinology & Diabetes & Metabolism.

We offer comprehensive support across all endocrinology & diabetology clinical trial phases, from Phase 1 to Phase 4, covering drug, biologic, and therapeutic development.
Clinical Endocrinology Trials are research studies testing new treatments for hormone and gland disorders like diabetes, thyroid, or obesity, ensuring safety, efficacy, and reliable patient outcomes.
These are research studies evaluating new therapies, devices, or interventions for hormonal and metabolic disorders, including diabetes, obesity, thyroid, adrenal, and pituitary conditions, to ensure safety, efficacy, and regulatory compliance.
Trials typically run in Phase I–IV, from early safety and dosing studies (Phase I) to large-scale efficacy trials (Phase III) and long-term post-marketing monitoring (Phase IV).
A CRO offers end-to-end support, including study design, regulatory submissions, site management, patient recruitment, data management, and pharmacovigilance, ensuring smooth and timely trial execution.
Common conditions include Type 1 & 2 diabetes, gestational diabetes, obesity, metabolic syndrome, thyroid disorders, adrenal and pituitary disorders, and diabetic complications like neuropathy or retinopathy.
Common endpoints include HbA1c, fasting glucose, weight/BMI changes, incidence of complications, and patient-reported outcomes, depending on the trial phase and study objectives.
Chronic disease trials include follow-up visits, lab tests, and patient-reported outcomes over months or years to evaluate sustained efficacy and monitor complications.
If you are planning to develop novel treatments in any therapeutic area, it’s time to consult a CRO that delivers rigor, efficiency, and regulatory clarity at every stage. Let’s position your therapy for global markets.

Be the first to know the latest trends in clinical research, real-world case studies, and industry secrets.

ProRelix Research is the rapidly growing Contract/ Clinical Research Organization (CRO) with multi-country service capability supporting phase 1, 2, 3, & 4 clinical trials of Pharma, Biotech, Biopharma, Medical Device, Nutraceutical & Herbal companies to conduct in the USA, India, Europe & South East Asia.