
eCOA in Clinical Trials ensures smarter clinical data collection, improved accuracy, patient compliance, and efficient trial outcomes.
What is eCOA in Clinical Trials?
eCOA in clinical trials are a type of clinical research in the field of healthcare and medicine. Electronic clinical outcome assessment (eCOA)refers to the use of electronic methods and technologies to collect patient-reported outcomes and other clinical data in a clinical trial setting. These electronic methods can include the use of smartphones, tablets, wearable devices, web-based platforms, or other digital tools to record and transmit information about a patient’s symptoms, experiences, and overall health status.
The primary goal of eCOA clinical trials is to improve the accuracy and efficiency of data collection during clinical research. By using electronic tools, researchers can reduce the potential for errors associated with manual data entry and streamline the data collection process, making it more convenient for both patients and investigators. eCOA methods are often used to assess patient-reported outcomes such as quality of life, symptom severity, and treatment-related side effects.
Significance of eCOA in Clinical Research
eCOA, or Electronic Clinical Outcome Assessment, has emerged as a key tool in modern clinical trials. eCOA in Clinical Trials offers numerous advantages, enhancing data accuracy and patient engagement, making it a fundamental aspect of contemporary research methodologies. By enabling patients to input their health-related information directly into electronic devices, eCOA minimizes transcription errors and streamlines data collection, ultimately improving the quality of trial outcomes. Furthermore, eCOA’s ability to facilitate remote data capture has become especially vital in the context of global health crises, ensuring trial continuity and participant safety. As we navigate the evolving landscape of clinical research,eCOA Clinical Trials remains at the forefront, revolutionizing the way we gather and utilize critical data.
The Advantages of eCOA in Clinical Trials over Traditional Paper-Based Data Collection Methods
The advantages of Electronic Clinical Outcome Assessment (eCOA) over traditional paper-based data collection methods presented in bullet points:
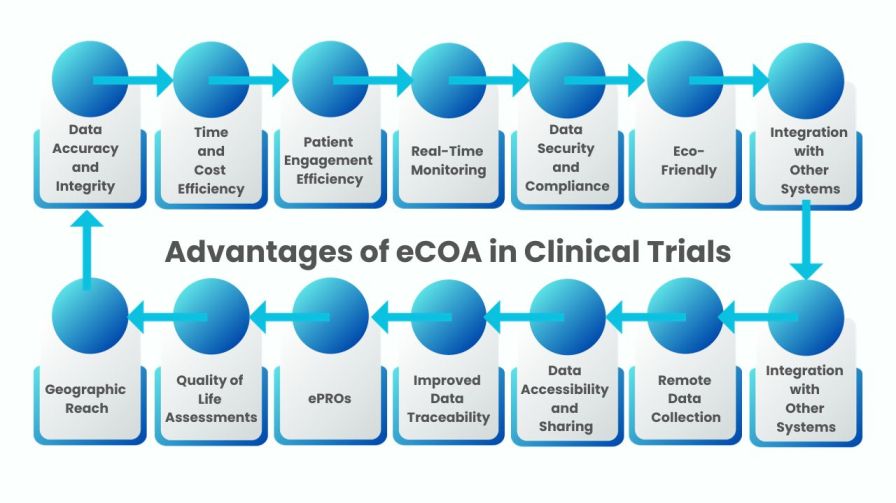
Figure: The Advantages of eCOA in Clinical Trials over Traditional Paper-Based Data Collection Methods
Data Accuracy and Integrity
- Reduced risk of transcription errors and data entry mistakes.
- Real-time data validation and automated skip patterns enhance data quality.
Time and Cost Efficiency
- Eliminates the need for printing, shipping, and storing paper forms.
- Faster data collection, review, and analysis, leading to shorter study timelines.
Patient Engagement
- User-friendly interfaces improve patient compliance and satisfaction.
- The ability to collect data remotely encourages broader patient participation.
Real-Time Monitoring
- Researchers can access data instantly, enabling prompt response to issues.
- Early identification of adverse events or protocol deviations.
Data Security and Compliance
- Enhanced data encryption and audit trails protect sensitive patient information.
- Adherence to regulatory standards (e.g., 21 CFR Part 11) is facilitated.
Eco-Friendly
- Reduces paper waste, contributing to a greener and more sustainable approach.
- Aligns with environmental responsibility goals.
Integration with Other Systems
- Easily integrates with Electronic Data Capture (EDC) and other clinical trial systems.
- Streamlines data flow and enhances the overall efficiency of clinical trials.
Remote Data Collection
- Allows for remote patient monitoring and data collection, reducing the need for physical site visits.
- Particularly valuable during global health crises (e.g., pandemics) to maintain data collection continuity.
Data Accessibility and Sharing
- Facilitates secure data sharing among stakeholders, including sponsors, CROs, and regulatory authorities.
- Enhances transparency in the research process.
Reduced Administrative Burden
- Minimizes the need for manual data handling, reducing administrative workload.
- Automates data collection and management tasks.
Customization and Adaptability
- Easily configurable to specific study requirements.
- Can accommodate a wide range of study types and data formats.
Improved Data Traceability
- Easy tracking of data changes and user interactions, aiding in the audit trail.
- Enhances the ability to reconstruct data history if needed.
Enhanced Patient-Reported Outcomes (ePROs)
- Supports the collection of patient-reported outcomes more efficiently and accurately.
- Provides a patient-centric approach to data collection.
Quality of Life Assessments
- Allows for real-time assessment of quality of life measures, particularly important in long-term studies or chronic disease research.
Geographic Reach
- Overcomes geographical barriers, enabling participation of patients from diverse locations.
- Facilitates multi-site and global clinical trials.
These advantages demonstrate how eCOA in clinical trials methods offer a significant improvement over traditional paper-based data collection in clinical research, leading to more efficient, reliable, and patient-centric outcomes.
How eCOA in Clinical Trials Improves Patient Engagement and Retention?
Electronic Clinical Outcome Assessments (eCOA) have Changed the way clinical trials are conducted, offering a range of benefits, including the enhancement of patient engagement and retention. Patient engagement and retention are critical factors in the success of clinical trials, as they directly Result the quality and reliability of the data collected. Here’s how eCOA achieves this
Convenience and Accessibility
eCOA platforms provide patients with the flexibility to complete assessments from the comfort of their own homes, reducing the need for in-person visits. This convenience makes it easier for patients to participate in the trial, especially those who may have mobility issues or live far from trial sites. As a result, patients are more likely to stay engaged throughout the trial’s duration.
Real-time Communication
eCOA systems enable real-time communication between patients and researchers. This means that patients can quickly report any issues or concerns, fostering a sense of being closely monitored and cared for. This transparency can improve patient satisfaction and trust in the clinical trial process.
User-Friendly Interfaces
eCOA platforms often come with user-friendly interfaces that are easy for patients to navigate. This reduces the burden on patients, making it less likely for them to become overwhelmed or frustrated with the assessment process. A positive user experience encourages patients to stay engaged in the trial.
Automated Reminders
eCOA in clinical trials systems can send automated reminders to patients, prompting them to complete assessments on time. This minimizes the risk of missed assessments and improves data reliability. Patients are more likely to remain engaged when they receive gentle reminders about their responsibilities in the trial.
Customization
eCOA platforms allow for the customization of assessments to better match a patient’s condition and individual needs. This personalized approach can make patients feel more valued, enhancing their commitment to the trial.
Data Visualization
Patients can often access their own assessment data through eCOA platforms, allowing them to track their progress. This visibility into their health data empowers patients and keeps them engaged in the study.
Reduced Burden
Paper-based assessments can be burdensome for patients, leading to fatigue and dropout. eCOA in clinical trials streamlines data collection and reduces the need for excessive paperwork. Patients are more likely to remain engaged when the process is simplified.
Safety and Compliance Checks
eCOA platforms can include built-in safety checks and validation rules, which ensure that patients provide accurate and complete data. This enhances data quality and compliance while giving patients a sense that their safety is a priority.
Data Security and Privacy in eCOA Clinical Trials
eCOA systems are designed to maintain the privacy and security of patient data. This assurance can make patients more comfortable with sharing their information, knowing that it will be handled with the utmost care.
Ensuring data security and privacy in electronic Clinical Outcome Assessments (eCOA) clinical trials is critical to safeguard patient information, maintain data integrity, and meet regulatory requirements. To achieve this, adherence to relevant data protection regulations, such as GDPR in Europe and HIPAA in the United States, is paramount. Robust encryption measures for data in transit and at rest should be in place to protect data from unauthorized access.
Strong user authentication, role-based access control, and audit trails help manage and monitor data access effectively. Data minimization practices, secure data storage, and encrypted data transmission are Important for protecting patient information. Furthermore, eCOA device security, oversight of third-party vendors, and informed consent processes are crucial to maintaining data security and patient privacy.
Well-defined data retention and disposal policies, along with a Complete data breach response plan, further enhance data protection. Regular training, awareness initiatives, and continuous monitoring ensure that data security remains a top priority throughout the eCOA in clinical trials process.
Data Integrity and Compliance in eCOA Clinical Trials
eCOA in clinical trials plays a critical role in ensuring data integrity and compliance in clinical trials. In an era where clinical research is becoming increasingly complex and data-driven, eCOA solutions have emerged as a powerful tool to enhance the quality of data collection, reduce errors, and streamline the regulatory compliance process.
Data Accuracy and Consistency
One of the primary benefits of eCOA is its ability to ensure the accuracy and consistency of data collected during clinical trials. By replacing paper-based assessments with electronic data capture methods, eCOA systems eliminate the risk of transcription errors, missing data, and illegible handwriting that can plague traditional data collection processes. This not only improves the quality of data but also minimizes the need for data cleaning, reducing the time and resources required for this tedious task.
Real-time Data Monitoring
eCOA systems enable real-time data monitoring, allowing sponsors and investigators to review and assess data as it is collected. This capability enhances the ability to detect discrepancies or irregularities promptly, facilitating timely corrective actions. In case of any issues or non-compliance with the trial protocol, immediate intervention is possible, preventing the accumulation of inaccuracies that could compromise the trial’s integrity.
Protocol Adherence
Clinical trial protocols are carefully designed to ensure patient safety, study integrity, and compliance with regulatory requirements. eCOA solutions can include built-in checks and validations to ensure that data collected aligns with the defined protocol. For instance, eCOA systems can flag entries that fall outside predefined ranges, require mandatory fields to be completed, or provide automated skip patterns based on participant responses. This promotes protocol adherence and minimizes deviations that could jeopardize data integrity.
Audit Trails and Data Security
Data integrity and compliance are closely related to data security. eCOA in clinical trials systems provide robust security measures to protect sensitive patient information and research data. Furthermore, these systems often maintain detailed audit trails that record every interaction with the data, including who accessed it, when, and what changes were made. This audit trail is invaluable for regulatory compliance and data integrity, as it allows complete traceability and accountability for all data-related actions.
Regulatory Compliance
eCOA solutions are designed with the latest regulatory guidelines and standards in mind, ensuring that clinical trials are conducted in accordance with Good Clinical Practice (GCP) and other relevant regulations. Data captured through eCOA is more likely to meet regulatory requirements due to its reduced risk of errors and the ability to implement necessary compliance checks. This minimizes the risk of non-compliance, which can result in regulatory sanctions or invalidated trial results.
Patient Engagement and Compliance in eCOA Clinical Trials
The user-friendly interfaces and remote data collection options of eCOA systems make it easier for participants to submit their data accurately and consistently. Patient compliance is crucial in maintaining data integrity, and eCOA can help by simplifying the data collection process for study participants.
The use of electronic methods for data collection in these trials, such as smartphones and web-based applications, not only simplifies the process for patients but also improves data accuracy through validation checks and real-time monitoring. eCOA systems can be designed with a patient-centric approach, offering flexible assessment times and automated reminders, reducing the burden on participants.
Remote monitoring and data collection enable decentralized trials and geographic diversity in recruitment. Privacy and security, compliance with data protection regulations, education, and training are crucial factors to ensure patient trust and effective use of eCOA. Additionally, the feedback loop facilitated by eCOA systems allows patients to report adverse events and concerns directly, contributing to quicker responses and improved patient safety. In eCOA clinical trials benefit significantly from enhanced patient engagement and compliance, ultimately leading to higher-quality data and study success.
Implementing eCOA in clinical trials can Improve data collection and enhance the overall efficiency of your research. Here are seven key considerations for successfully implementing eCOA in clinical trials:
Regulatory Compliance
Ensure that your eCOA system complies with the regulatory requirements of the regions where your clinical trials are conducted. Familiarize yourself with guidelines such as 21 CFR Part 11 in the United States and similar regulations in other countries.
Device Compatibility
Consider the various devices that participants may use to enter data, such as smartphones, tablets, or web browsers. Ensure that your eCOA system is compatible with these devices, taking into account factors like screen size and operating systems.
Patient Engagement
Engage with patients early in the trial planning process to ensure their comfort and willingness to use the eCOA system. Provide training and support to help participants become familiar with the technology.
Data Security
Prioritize the security of patient data. Implement robust data encryption, user authentication, and access controls to safeguard sensitive information. Regularly update security protocols to stay ahead of emerging threats.
Data Validation and Quality Assurance
Establish rigorous data validation and quality assurance processes. These should include edit checks, audit trails, and monitoring for missing or inconsistent data. Ensuring data integrity is crucial for the credibility of your trial results.
Integration with Other Systems
Consider how the eCOA system will integrate with other trial-related software, such as electronic data capture (EDC) systems and ePRO (electronic patient-reported outcomes) databases. Seamless integration can reduce data transfer errors and streamline trial management.
By addressing these considerations, you can maximize the benefits of eCOA in your clinical trials, such as improved data accuracy, increased patient engagement, and enhanced trial efficiency. However, it’s Important to adapt your approach to the specific needs and circumstances of your trial and continuously monitor and refine your eCOA in clinical trials implementation throughout the study.
How eCOA Is Used in Cancer Studies to Improve Clinical Trial Outcomes?
Electronic Clinical Outcome Assessment (eCOA) is increasingly used in cancer studies to capture accurate, real-time patient data throughout clinical trials. In oncology research, eCOA enables patients to directly report symptoms, side effects, pain levels, quality of life, and treatment tolerability using digital devices such as smartphones, tablets, or web platforms. This approach reduces data entry errors, eliminates paper-based delays, and enhances compliance by sending automated reminders. In cancer trials, where patient-reported outcomes are critical for evaluating treatment effectiveness and safety, eCOA in clinical trials improves data reliability and supports faster regulatory submissions. By enabling remote data collection, it also reduces patient burden—an important factor for individuals undergoing intensive cancer treatments—ultimately contributing to more patient-centric and efficient oncology clinical research.
Why eCOA Is Transforming Clinical Research?
ectronic Clinical Outcome Assessment (eCOA) is transforming clinical research by modernizing how patient data is collected, managed, and analyzed. Unlike traditional paper-based assessments, eCOA enables real-time data capture through digital devices such as smartphones, tablets, and web platforms. This significantly reduces manual errors, eliminates missing entries, and ensures higher data accuracy through built-in validation checks. With instant data availability, sponsors and CROs can monitor trial progress more efficiently and identify potential safety or compliance issues at an early stage.
eCOA also enhances patient engagement and compliance by offering automated reminders and user-friendly interfaces, leading to improved completion rates. Its flexibility supports decentralized and hybrid clinical trial models by allowing patients to report outcomes remotely, reducing the need for frequent site visits and improving overall participant convenience. Furthermore, regulatory authorities such as the U.S. Food and Drug Administration recognize and provide guidance on the use of electronic systems in clinical trials, reinforcing the credibility and acceptance of eCOA solutions.
By accelerating data cleaning, shortening database lock timelines, and improving operational efficiency, eCOA contributes to faster study completion and cost optimization. As clinical trials continue to evolve toward more patient-centric and technology-driven approaches, eCOA has become a critical component in delivering high-quality, reliable, and timely clinical research outcomes.
eCOA Clinical Trials Future Trends and Innovations
eCOA (Electronic Clinical Outcome Assessment) is rapidly shaping the future of clinical trials. This innovative technology is revolutionizing data collection by streamlining the process, enhancing patient engagement, and improving data quality and integrity. With the increased adoption of eCOA clinical study, we can expect a shift towards more patient-centric approaches, real-time data monitoring, and a reduction in human errors. These advancements hold the potential to significantly enhance the overall success and efficiency of clinical trials, ultimately benefiting both patients and researchers.
Future trends and Progress in the field of eCOA in clinical trials:
Digital Biomarkers Integration
The integration of digital biomarkers in eCOA (Electronic Clinical Outcome Assessments) is expected to become a significant trend. These biomarkers, collected from wearable devices and mobile apps, can provide real-time, objective data on patients’ health and well-being, enhancing the quality and accuracy of clinical trial assessments.
Patient-Centric eCOA Design
Future eCOA solutions will prioritize patient-centric design, making it easier for patients to participate in clinical trials. This includes user-friendly interfaces, multilingual options, and customizable assessments to ensure patients’ diverse needs are met.
Real-World Evidence (RWE) Integration
To streamline drug development, eCOA systems will increasingly incorporate real-world evidence from electronic health records and other sources. This data can provide a more Complete view of a drug’s effectiveness and safety profile in real-world settings.
AI and Machine Learning for Data Analysis
Artificial intelligence and machine learning algorithms will play a vital role in eCOA data analysis. These technologies can identify subtle trends, predict patient outcomes, and enhance the efficiency of clinical trials by automating data quality control and anomaly detection.
Remote Monitoring and Telehealth
The COVID-19 pandemic accelerated the adoption of remote monitoring and telehealth solutions in clinical trials. eCOA in clinical trials systems will continue to support virtual patient visits, enabling real-time data collection, reducing the need for in-person visits, and increasing patient retention rates.
Blockchain for Data Security
Given the sensitivity of clinical trial data, blockchain technology is expected to be increasingly employed to ensure the security, integrity, and traceability of eCOA data. This will enhance data privacy, reduce fraud, and improve transparency in clinical trial operations.
Regulatory Advances
As eCOA systems become more sophisticated and widely adopted, regulatory bodies like the FDA will likely provide clearer guidance and regulations to ensure data quality, patient privacy, and the validity of eCOA in clinical trials. Staying informed and compliant with these evolving regulations will be crucial for sponsors and Clinical Research Organizations.
These trends and innovations in the field of eCOA for clinical trials are expected to reshape the way trials are conducted, making them more patient-centric, data-driven, and efficient. It’s crucial for stakeholders in the pharmaceutical and clinical research industries to stay updated with these developments to remain competitive and ensure the successful execution of clinical trials.
Take on the Control of eCOA to Breakthrough Your Clinical Trial Data Collection Process
Take on the control of eCOA to Change Your Clinical Trial Data Collection Process. In today’s rapidly evolving landscape, hybrid clinical trials have become a pivotal component in drug development. Leveraging Electronic Clinical Outcome Assessments (eCOA) can significantly enhance the efficiency and accuracy of data collection in these trials. By incorporating eCOA solutions, you can seamlessly capture patient-reported outcomes and clinical data, ensuring a streamlined and patient-centric approach that optimizes the success of your hybrid clinical trials. The impact of eCOA on data quality and integrity is profound, offering real-time, accurate, and patient-centric data collection that enhances the reliability of clinical trial results. By replacing traditional paper-based methods with digital, patient-friendly platforms, eCOA in clinical trials streamlines data collection, reduces errors, and ultimately contributes to more efficient and successful clinical trials.
What is the difference between eCOA and ePRO clinical trials?
In the context of clinical trials, eCOA and ePRO serve as complementary yet distinct tools for gathering critical outcome data, yet they differ both in breadth and in the specific perspectives they emphasize. eCOA constitutes an umbrella framework for any clinical outcome acquired electronically, encompassing submissions from multiple informants—patients, clinicians, trained observers, and performance-rating exercises. Within eCOA, PROs, ClinROs, ObsROs, and performance data (PerfOs) are collected under standardized electronic conditions. Such inclusiveness allows stakeholders to triangulate treatment effects using multiple evaluative lenses (clinical, patient-generated, and observer) and to build a richer, multi-dimensional picture of a patient’s evolving status.
Conversely, ePRO represents the patient-facing portion of the wider eCOA portfolio and aims to collect data solely from the patient’s own perspective. Whether through a smartphone questionnaire, tablet diary, or a browser-based tool, patients electronically submit correspondence on symptoms, daily functioning, and the extent of treatment adherence, all recorded without intermediary involvement. The ePRO methodology safeguards the authenticity of the patient’s report by minimizing external influence and reflective delay, thereby ensuring that frequent, self-reported clinical impressions are captured and transmitted in real time.
The principal distinction between eCOA ePRO stems from data origin and Completeness. ePRO captures exclusively what patients report themselves, yielding focused insights into their lived experiences. eCOA expands on this framework by also incorporating observations recorded by clinicians and objective performance metrics, resulting in a rounded view that bolsters the evidentiary base of a clinical trial. In practice, ePRO’s narrower, straightforward focus makes data collection user-friendly and firmly patient-centered. In contrast, eCOA’s wider analytical scope is inherently more intricate, drawing on multiple data types to paint a richer clinical picture. By recognizing these differences, sponsors can select the optimal modality based on the study’s design objectives, choosing a streamlined ePRO clinical trials collection when patient perception is paramount or eCOA when integrating heterogeneous domain assessments is required for Complete assessment.
eCOA vs ePRO
|
ePRO (electronic Patient-Reported Outcomes) |
eCOA (electronic Clinical Outcome Assessment) | |
|
Definition |
Data provided directly by patients about their health status, without any interpretation by healthcare professionals or others. |
A broader category that encompasses ePRO as well as clinician-reported, observer-reported, and performance outcomes. |
|
Source of Data |
Patients |
Patients, clinicians, caregivers, or via performance tests. |
|
Scope |
Centered on the patient’s personal experience, such as symptoms, quality of life, and adherence to treatment. |
Includes all types of outcome assessments in trials, such as ePRO, eClinRO, eObsRO, and ePerfO. |
|
Examples |
Pain level, fatigue, mood, daily activity limitations. |
ePRO (patient diary), eClinRO (physician assessment), eObsRO (parent/caregiver notes), ePerfO (cognitive/motor tests). |
|
Use in Clinical Trials |
Directly captures the patient’s perspective, which is crucial for patient-centered outcomes. | Delivers a Complete outcome assessment framework encompassing multiple perspectives. |
|
Technology |
Mobile apps, tablets, web portals, wearables used by patients. |
Includes the same tools as ePRO plus clinician input systems and performance testing tools. |
|
Regulatory Acceptance |
Highly recommended by the FDA and EMA for capturing patient-centered outcomes. |
Acknowledged as the gold standard for standardized data collection in clinical trials. |
eCOA Benefits in Clinical Trials: Complete 2026 Guide
Electronic Clinical Outcome Assessment (eCOA) is transforming clinical trials in 2026 by replacing paper-based assessments with secure digital platforms such as mobile apps and web-based tools. It enables the collection of patient-reported, clinician-reported, observer-reported, and performance outcomes in real time. One of the key benefits of eCOA is improved data accuracy and quality, as automated validations reduce errors, missing data, and recall bias. These systems also support regulatory compliance with agencies like the U.S. Food and Drug Administration and the European Medicines Agency through audit trails and secure data handling.
eCOA also enhances patient engagement by allowing remote participation, reducing site visits, and improving protocol adherence through automated reminders. For sponsors and CROs, this leads to faster data access, efficient monitoring, reduced operational costs, and quicker study timelines. As decentralized and hybrid trials continue to grow, eCOA plays a crucial role in improving data integrity, patient experience, and overall trial efficiency.
Closing Thoughts
eCOA (electronic Clinical Outcome Assessment) has undeniably Came out as a transitioner in the realm of clinical trials, Modernizingthe way data is collected and transforming the entire research landscape. With its numerous advantages, including increased data accuracy, patient engagement, and cost-efficiency, eCOA in clinical trials has certified to be an indispensable tool for researchers and pharmaceutical companies alike. As the clinical trials industry continues to evolve, embracing eCOA is not just a choice but a necessity to Improve the quality and efficiency of data collection, ultimately expediting the development of innovative therapies and improving patient outcomes. In a world driven by technology andinnovation, eCOA in clinical trials has unquestionably earned its place as akey keyword in the pursuit offrontline research and healthcare advancements Progress.
FAQ’S
What is the purpose of the eCOA system?
The purpose of an eCOA in clinical trials system was set up to collect respondents,including the patient, the clinician, or the caregiver proprietary outcome data during clinical trials while maintaining precision to avoid errors and enhance compliance. It enables remote data collection that is real-time, standardized, and in accordance with legal regulations, provides remote participation, and optimizes the overall efficiency and data integrity of the entire trial.
Why is eCOA important?
The use of eCOA greatly streamlines the clinical trial process. Entering clinical data electronically mitigates patient, clinician, or outcome assessor errors, eliminates missing data, shifts data collection from cross-sectional to streaming methodologies, and streamlines errors and compliance to critical “must-have” clinical trial requirements. Quality of patient involvement, data collected, and the resultant clinical trial are all achieved to higher standards.
What is the eCOA review?
eCOA review is an important step towards verifying data derived from an electronic Clinical Outcome Assessment system during the trial phase of a study. It reviews while ensuring data accuracy is Complete all outcomes reported by patients, clinicians, and other observers are scrutinized, and anomalies of absence or insertion are detected and validated against the prescribed criteria of entry protocols. This review ensures the quality of data, data is compiled and stored, analysis is conducted, and the findings are submitted. It also contributes to compliance and regulatory preparedness, enabling real time analytics for the sponsors and CRO’s.
What does eCOA stands for in clinical trials?
The eCOA full form in Clinical Trials is Electronic Clinical Outcome Assessment. It refers to the use of digital devices such as smartphones, tablets, or web-based platforms to electronically capture clinical trial data directly from patients, clinicians, or caregivers. eCOA in clinical trials improves data accuracy, enhances patient compliance, enables real-time monitoring, and reduces errors compared to traditional paper-based methods, making it an essential component of modern and decentralized clinical trials.
What is the difference between ePRO and eCOA?
ePRO concerns patients recording their own level of health, symptoms, or quality of life electronically. eCOA refers to ePRO plus clinician-reported outcome (ClinRO) assessments, observer-reported outcome (ObsRO) assessments, and performance-based outcome (PerfO) assessments. Thus, ePRO is more limited than eCOA, which has diverse clinical outcome reporting.
What is ePRO in clinical trials?
ePRO refers to electronic Patient-Reported Outcomes, which involve the automated collection of patient-reported information on symptoms, side effects, quality of life, and overall treatment experience through electronic platforms such as mobile phones, tablets, and web portals. This method improves data accuracy, reduces missing information, and enables continuous monitoring. Recognized by global regulatory authorities as a reliable data collection approach, ePRO offers benefits such as improved data completeness and quality, greater convenience for patients and healthcare providers, and enhanced responsiveness from providers.




